What is the meaning of latent energy?

Andrew Mclaughlin
Published Mar 17, 2026
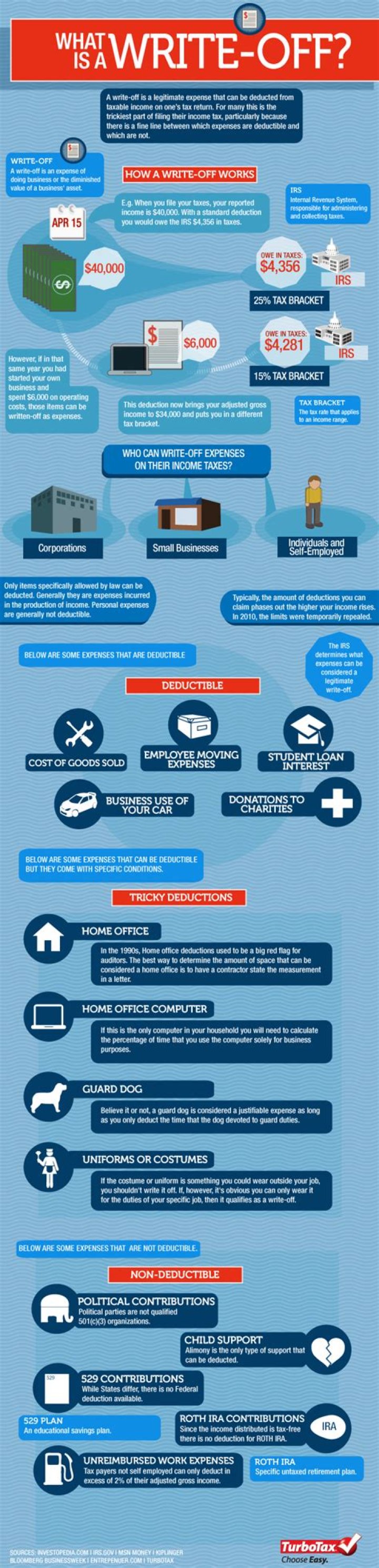
Latent heat (also known as latent energy or heat of transformation) is energy released or absorbed, by a body or a thermodynamic system, during a constant-temperature process — usually a first-order phase transition.
What is latent heat of energy?
Latent heat, energy absorbed or released by a substance during a change in its physical state (phase) that occurs without changing its temperature. The latent heat is normally expressed as the amount of heat (in units of joules or calories) per mole or unit mass of the substance undergoing a change of state.
What is latent heat of fission?
The amount of heat energy exchanged per unit mass of a substance is called Latent Heat. Latent heat of fission is analogous to latent heat of fusion.
What causes latent heat?
All pure substances in nature are able to change their state. Solids can become liquids (ice to water) and liquids can become gases (water to vapor) but changes such as these require the addition or removal of heat. The heat that causes these changes is called latent heat.
Is latent heat of fusion constant?
The ‘enthalpy’ of fusion is a latent heat, because, while melting, the heat energy needed to change the substance from solid to liquid at atmospheric pressure is latent heat of fusion, as the temperature remains constant during the process. Once the water is completely frozen, its temperature continues to fall.
Does latent heat change with pressure?
As pressure increases, pressure acting helps in binding the molecules thus even removal of lesser amount heat would also do. Thus as presure increases at 100 degree latent heat of vapourisation also increases while as pressure increases latent heat of condensation decreases.
What are the four types of latent heat?
Types of Latent Heat
- Latent Heat of Fusion,
- Latent Heat of Vaporization,
- Latent Heat of Sublimation.
What is the latent heat for melting?
A total of 334 J of energy are required to melt 1 g of ice at 0°C, which is called the latent heat of melting. At 0°C, liquid water has 334 J g−1 more energy than ice at the same temperature. This energy is released when the liquid water subsequently freezes, and it is called the latent heat of fusion.
How many latent heat can a substance have?
As there can be two boundaries for change, solid/liquid and liquid/gas, each material has two specific latent heats: latent heat of fusion – the amount of energy needed to freeze or melt the material at its melting point.
Can you feel latent heat?
However, although you cannot feel it, the liquid has stored all that latent heat. The only way you will observe the latent heat is if you try to transform the water back to ice. Heat energy is conserved no matter how the phase change occurs. If you put heat into water, it can evaporate.
What is latent heat of fusion with example?
For example, the latent heat of fusion of one kilogram of water, which is the amount of heat energy that must be supplied to convert 1 kg of ice without changing the temperature of the environment (which is kept at zero degrees celsius) is 333.55 kilojoules.
What is specific latent heat?
The specific latent heat of a substance is the amount of energy needed to change the state of 1 kg of the substance without changing its temperature. Each substance has two specific latent heats: latent heat of fusion (the amount of energy needed to freeze or melt the substance at its melting point)
What process absorbs the most latent heat?
An example is latent heat of fusion for a phase change, melting, at a specified temperature and pressure. Latent heat is associated with the phase changes of atmospheric water vapor, mostly vaporization and sublimation.
Why does the latent heat of steam decreases as we increase the pressure?
The heat energy (enthalpy of evaporation) needed by the water at higher pressure to change it into steam is actually less than the heat energy required at atmospheric pressure. This is because the specific enthalpy of evaporation decreases as the steam pressure increases.
Is latent heat potential energy?
Latent heat is a form of internal or potential energy stored by evaporated or melted water. As ice melts or liquid water evaporates, the molecules change state — from a solid to a liquid, from a liquid to a gas, or from a solid directly to a gas.
What are the four different types of latent heat?
Is Sweating an example of latent heat?
Well, when you sweat, this is a very clear example for Sensible and Latent heat interchange. When you do any effort you sweat, which is basically water on the surface of your skin. That’s why in humid climates when sweat doesn’t evaporate that fast because the climate is Saturated with water you feel hotter.
What is the biggest difference between sensible and latent heat?
Sensible heat is the heat that causes an object to change temperature. However, latent heat does not affect the temperature of a substance or object. Water for example boils at 100°C and the latent heat keeps the water boiling. Total capacity in an air conditioner is the sum of the sensible and latent heat values.
What are three types of latent heat?
There are three different types of latent heats,
- Latent Heat of Fusion,
- Latent Heat of Vaporization,
- Latent Heat of Sublimation.
Latent heat, energy absorbed or released by a substance during a change in its physical state (phase) that occurs without changing its temperature.
What is sensible and latent energy?
Latent and sensible heat are types of energy released or absorbed in the atmosphere. Latent heat is related to changes in phase between liquids, gases, and solids. Sensible heat is related to changes in temperature of a gas or object with no change in phase.
Latent heat of vaporization is a physical property of a substance. It is defined as the heat required to change one mole of liquid at its boiling point under standard atmospheric pressure. It is expressed as kg/mol or kJ/kg.
Why is latent heat important?
That is latent heat. When the molecules return to a liquid state the latent heat is released as sensible heat (heat you can feel basically). It is important in the atmosphere because it is the primary factor involved in the formation of convective clouds and the stability/instability of the atmosphere.
What is the importance of latent heat?
Why do we use latent heat?
The latent heat of fusion is the heat required for an object to go from the solid state to the liquid state, or vice versa. Since its value is generally much higher than specific heat, it allows you to keep a beverage cold for much longer by adding ice than simply having a cold liquid to begin with.
What is latent capacity?
Latent Capacity is the unrealised production of existing operations that achieve increased production output with minimal investment.
What is the meaning of latent energy in chemistry?
Latent energy, or sometimes latent heat, represents the energy required to separate or break bonds in molecules or substances.
How is latent heat related to enthalpy property?
Latent Heat Latent heat is defined as the heat or energy that is absorbed or released during a phase change of a substance. It could either be from a gas to a liquid or liquid to solid and vice versa. Latent heat is related to a heat property called enthalpy.
How is latent heat related to phase change?
Latent heat is the heat the results from an increase or decrease in the amount of moisture held by the air. Specifically, it’s the amount of energy needed to cause a phase change (for our purposes, liquid-to-gas or gas-to-liquid) without changing the actual temperature of a substance.
What’s the difference between latent heat and sensible heat?
Specifically, it’s the amount of energy needed to cause a phase change (for our purposes, liquid-to-gas or gas-to-liquid) without changing the actual temperature of a substance. Humidity itself isn’t latent heat, but humidity contains latent heat.