How are drug trials carried out?

James Williams
Published Feb 13, 2026
Most clinical trials use comparison groups to compare medical strategies and treatments. Results will show if one group has a better outcome than the other. This is usually conducted in one of two ways: One group receives an existing treatment for a condition, and the second group receives a new treatment.
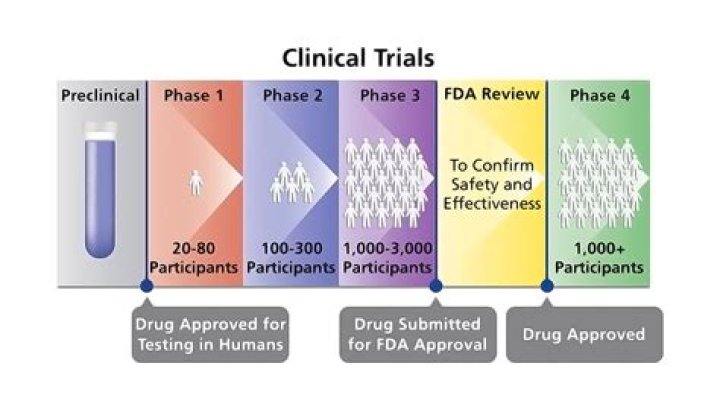
What are the 4 phases of a clinical trial?
Summary
| Phase | Primary goal |
|---|---|
| Phase I | Dose-ranging on healthy volunteers for safety |
| Phase II | Testing of drug on participants to assess efficacy and side effects |
| Phase III | Testing of drug on participants to assess efficacy, effectiveness and safety |
| Phase IV | Post marketing surveillance in public |
How much do you get paid to test drugs?
Each year, US scientists require a total of at least 10 million healthy test subjects, says Adil Shamoo, chair of Circare, a human research watchdog organization. Depending on duration, rigor, and risk, medical studies can pay as much as $10,000 each. The work isn’t demanding, but it can be dangerous.
What is a Phase 3 drug trial?
Phase III clinical trials compare the safety and effectiveness of the new treatment against the current standard treatment. Because doctors do not yet know which treatment is better, study participants are often picked at random (called randomized) to get either the standard treatment or the new treatment.
Who runs a clinical trial?
Who Conducts Clinical Studies? Every clinical study is led by a principal investigator, who is often a medical doctor. Clinical studies also have a research team that may include doctors, nurses, social workers, and other health care professionals.
What is a Phase 4 study?
A type of clinical trial that studies the side effects caused over time by a new treatment after it has been approved and is on the market. Phase IV clinical trials may include thousands of people. Also called phase 4 clinical trial and post-marketing surveillance trial.
How many stages of clinical trials are there?
There are 3 main phases of clinical trials – phases 1 to 3. Phase 1 trials are the earliest phase trials and phase 3 are later phase trials. Some trials have an earlier stage called phase 0, and there are some phase 4 trials done after a drug has been licensed.
Is it safe to pass a drug test?
The net is deliberately wide so they can catch any offender that may slip through the cracks. So as long as you don’t give them any reason to suspect that you’re using drugs, you should be safe from any kind of drug testing.
What happens when you take a drug test?
In short, urine drug tests can span an array of different testing methods. You should never think that it’s over once you hand in your urine sample. They can put you under observation, ask you to take another kind of drug test (more on that later), or even have you take two urine tests one after the other. When Will I Be Asked to Take a Drug Test?
Can you make passing a drug test a condition of employment?
You can make passing a drug test a condition of employment. With this approach, all job candidates will receive drug testing prior to being hired. You can test your employees for alcohol and other drug use as part of an annual physical examination. Be sure to inform employees that drug-testing will be part of the exam.
How are new drugs tested in the laboratory?
New medical drugs have to be tested to ensure that they work, and are safe, before they can be prescribed. The drugs are tested using computer models and human cells grown in the laboratory. Many substances fail this test because they damage cells or do not seem to work. Drugs that pass the first stage are tested on animals.



